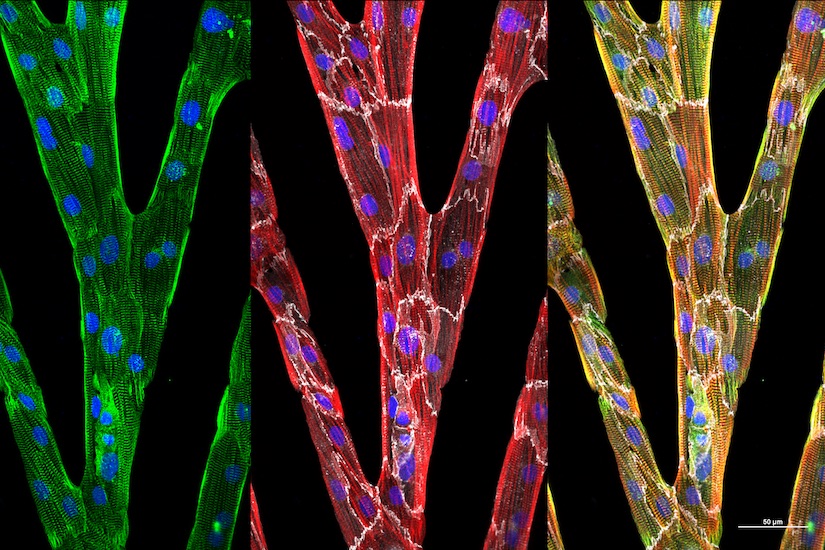
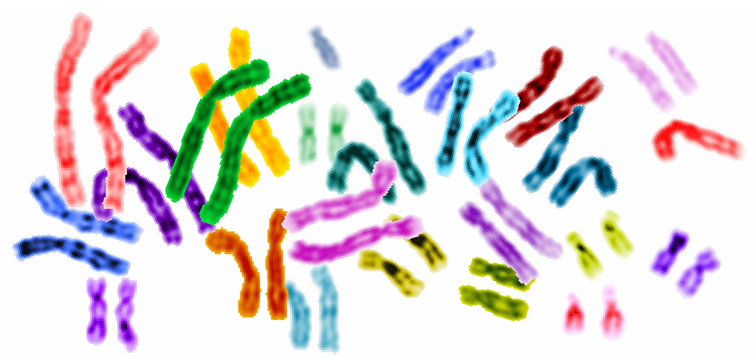
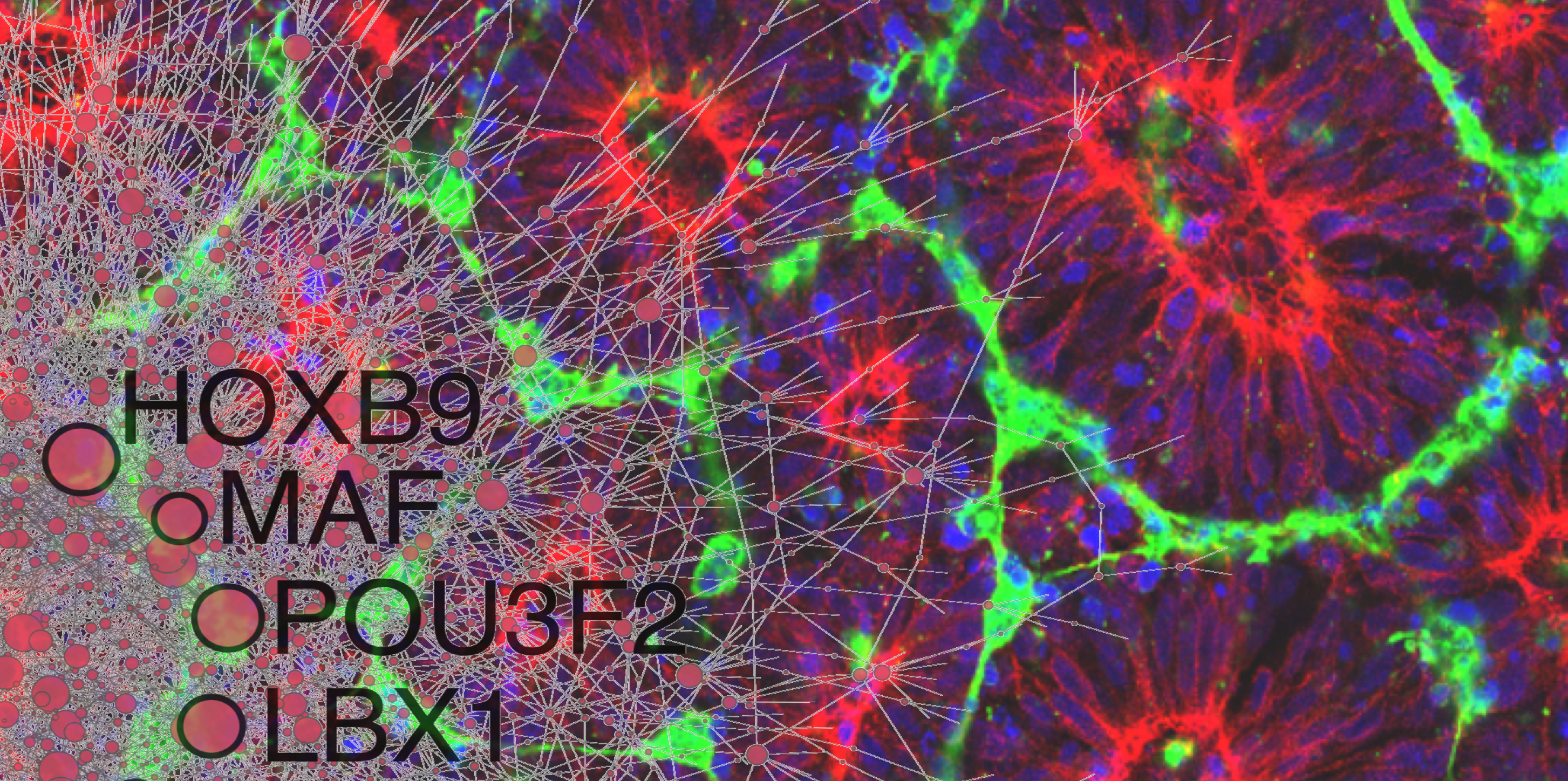
Precision medicine is an emerging approach for disease treatment and prevention that takes into account individual variability in genes, environment, and lifestyle for each person. This approach will allow doctors and researchers to predict more accurately which treatment and prevention strategies for a particular disease will work in which groups of people. It is in contrast to a “one-size-fits-all” approach, in which disease treatment and prevention strategies are developed for the average person, with less consideration of the differences between individuals.
Researchers at WID with expertise in complex biological systems, epigenetics, data science, health care, gene editing, and tissue engineering will contribute to “healthy people” initiatives through precison medicine.
Precision medicine is an important component of WID’s Multi-Omics Hub.