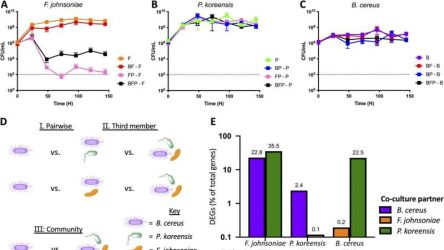
Serendipity reveals new method to fight cancer with T cells
Cells treated with “metabolic priming” retained their stem cell-like qualities, thus enhancing their ability to kill cancer cells, transform into durable memory cells, and survive longer in the body.